NMN Powder: Pure NMN Supplement for Health and Longevity
Pure NMN Powder is a pharmaceutical-grade nucleotide that helps make NAD+, an important coenzyme for DNA repair and cell energy production. Unlike most dietary supplements, this bioactive molecule is more than 99% pure, isolating the Beta-NMN anomer and getting rid of forms that aren't biologically active. By targeting important cellular ageing pathways, it is a key ingredient in nutraceutical formulations that aim to improve longevity and vitality in the global health supplement market. The growing interest in compounds that boost NAD+ has changed how we think about cellular health and ageing. As a B2B ingredient expert who works with formulation teams and procurement managers in the nutraceutical industry, I've seen how nicotinamide mononucleotide has become an important raw material for making products that aim to make people live longer. This detailed guide looks at the technical aspects, sourcing issues, and practical uses that mean most to professionals who want to add high-quality NAD+ precursors to their product lines.
Understanding Pure NMN Powder – Science and Benefits
The biochemical foundation of nicotinamide mononucleotide centres on its role as an immediate NAD+ precursor within human cellular metabolism. When we examine cellular aging at the molecular level, declining NAD+ concentrations emerge as a central factor affecting mitochondrial function, DNA repair capacity, and overall cellular vitality. Research published in leading journals demonstrates that NAD+ levels decrease by approximately 50% between the ages of 40 and 60, creating a metabolic bottleneck that impacts numerous physiological processes.
Chemical Composition and Molecular Structure
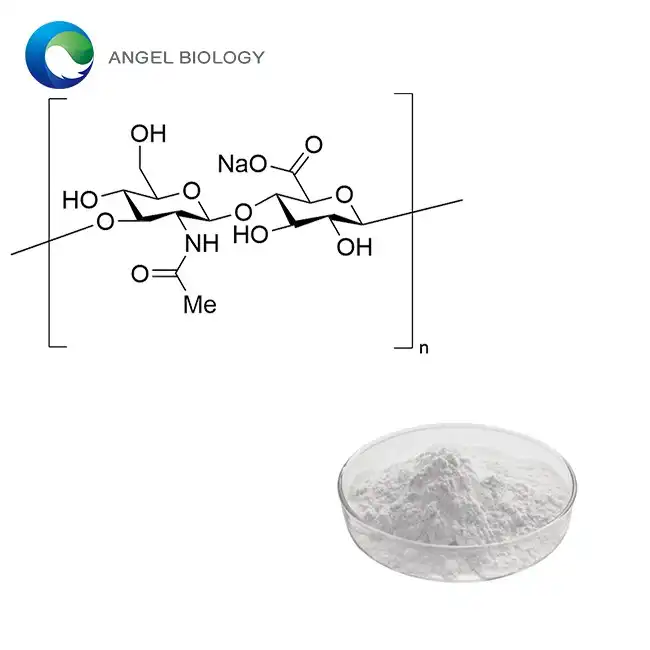
With a molecular weight of 334.22 g/mol, the molecular formula C11H15N2O8P tells us what this nucleotide compound is. High-Performance Liquid Chromatography testing shows that the assay is more than 99% pure, which sets pharmaceutical-grade material apart from lower-quality options. The Beta-anomer configuration works naturally, but the Alpha-forms are not useful for therapy. Modern enzymatic catalysis production methods make sure that the stereochemistry is pure, and they get rid of the harmful solvent leftovers that are often left over from chemical synthesis routes. This way of making things solves problems that the industry has had in the past with yield efficiency and cost-effectiveness while still meeting the highest quality standards. The crystalline structure shows specific optical rotation values between -15° and -20° when tested under normal conditions, which proves that the stereochemistry is correct. During industrial audits, this technical parameter is a very important quality control checkpoint. Bulk density readings between 0.5 and 0.7 g/mL show the best powder properties for further processing in uses like capsule filling and tablet compression.
NAD+ Biosynthesis Pathway and Cellular Mechanism
Nicotinamide mononucleotide gets into human cells through the salvage route, skipping steps that slow down other precursor compounds for enzymes. The process of change happens quickly in peripheral tissues, where special transporters make it easier for cells to take in the material. Phosphorylation changes the molecule into NAD+ inside cells with the help of NMN adenylyltransferase enzymes. It is this efficient metabolic pathway that explains why this precursor is more bioavailable than others that have been studied. In mammals, the salvage pathway is the main way that NAD+ homeostasis is maintained. Unlike starting from scratch with tryptophan, this recycling method works very well with compounds that contain nicotinamide. Clinical findings show that taking it by mouth leads to a measurable increase in NAD+ levels in the liver and skeletal muscles within hours. Pharmacokinetic studies show that peak plasma concentrations happen about 30 minutes after administration, and that accumulation patterns change depending on the expression profiles of transporters.
Scientifically Validated Health Applications

A lot of studies using different model systems have found a few main biological effects connected with NAD+ restoration. Bioenergetics in mitochondria get better when the electron transport chain works better, and there is enough NAD+ available. Enzymes called PARPs, which use NAD+, are very important for DNA repair because they find and fix genetic damage. Another group of NAD+-dependent enzymes, called sirtuin proteins, control metabolic adaptation and stress resistance pathways all over the body. Dosage ranges from 250 mg to 1000 mg daily have been tested in human clinical studies, and safety profiles have remained good at all of these levels. Biomarker studies show that following dosing for 4 to 12 weeks, there are measurable increases in blood NAD+ metabolites. These results give formulation teams the proof they need to make products for people who want to improve cellular health and good ageing. The fact that there were no big adverse events in published Pure NMN Powder trials is reassuring for regulatory compliance in major markets.
Comparative Analysis of Pure NMN Powder and Alternative Supplements
Procurement decisions in the longevity supplement category require careful evaluation of competing ingredient options. The landscape includes various nicotinamide derivatives, each with distinct chemical properties, absorption characteristics, and manufacturing considerations. Understanding these technical differences helps buyers align ingredient selection with specific product positioning and target consumer preferences.
Purity and Bioavailability Considerations
Advanced production methods make it possible for pharmaceutical-grade nicotinamide mononucleotide to have purity levels higher than 99.5%. This is very different from lower-grade materials, which might have large amounts of inactive anomers, degradation products, or be manufactured by-products. High-density crystalline forms don't break down when they come in contact with water, so they stay effective all the way through the supply chain, from making to the end user. The engineered crystal lattice structure limits the amount of surface area that is exposed to moisture, which stops the hydrolysis reactions that turn the active compound into nicotinamide and give off unpleasant smells. Absorption studies that compare different NAD+ precursors show that their bioavailability profiles are significantly different. Nicotinamide mononucleotide can enter cells directly through certain transporters, but nicotinamide riboside needs to be phosphorylated inside cells before it can start the NAD+ synthesis route. Because of this difference in metabolism, the drugs are distributed differently in tissues and follow different pharmacokinetic trends. Formulation scientists use these traits to help them make goods that work well in certain situations, like when they need to have a quick effect or a long-lasting effect.
Format Options and Manufacturing Compatibility
In this group of ingredients are crystalline powders, capsule mixes, and ready-to-use formulations, among other forms. The raw powder form gives you the most freedom when making your own formulations because it lets you test exact dosing and compatibility with other active ingredients. Encapsulated goods are easier to handle for brands that want turnkey solutions, but they don't let you change the way they're made as much. Sublingual tablets are another form meant to improve absorption in the mouth, but they are harder to make and need to be carefully thought out. When it comes to making, the way powder is handled has a big effect on how efficiently it is made. High-density variants constantly flow through equipment that fills capsules, which cuts down on waste and boosts throughput. Low-density forms work well for uses that need to dissolve quickly and become hydrated quickly. These technical factors have a direct effect on the prices of production and the consistency of quality across batches of production. When reviewing supplier proposals, procurement teams should ask for detailed powder specification sheets that show how the particles are distributed in size, the bulk density, and the moisture content.
Cost-Effectiveness and Market Positioning
Nicotinamide mononucleotide prices depend on a lot of things, like how it is made, the purity grade, whether it is certified, and the number of orders that come in. Enzymatic synthesis costs more than chemical synthesis methods because it produces better quality profiles and is better for the environment. Most of the time, bulk buy agreements let you get volume discounts. However, smaller brands or new products may have trouble meeting the minimum order quantities. Smart buying plans weigh the cost per unit against the need to ensure quality and the need to keep an eye on inventory. A study of the market shows that people are becoming more aware of how to make sure ingredients are pure and that production processes are open. Clean-label trends favour products made with biotechnology over chemical synthesis. This is in line with customer preferences for natural and environmentally friendly production methods. This way the market works supports charging more for ingredients that are clearly of better quality. This lets brands stand out by focusing on quality instead of just competing on price. Strategic buyers know that the quality of the ingredients has a long-term effect on how well a product works, how happy customers are with it, and how well the brand is known.
How to Source and Procure Pure NMN Powder Efficiently
Identifying reliable manufacturing partners represents Pure NMN Powder perhaps the most critical decision in the procurement process. The global supply landscape includes producers ranging from small-scale chemical suppliers to vertically integrated biotechnology companies with dedicated research facilities. Distinguishing between these options requires systematic evaluation frameworks and a clear understanding of quality benchmarks.
Evaluating Manufacturer Credentials and Certifications
Good Manufacturing Practice (GMP) certification is the minimum quality level for ingredient suppliers who mean business. GMP compliance means that strong quality management systems have been put in place to check the raw materials, keep an eye on the process, keep track of each batch, and test the final product. In addition to basic GMP, you should look for suppliers who are certified in both ISO 9001 (for quality management) and ISO 22000 (for food safety systems). These international standards show that an organisation is dedicated to ensuring quality in all areas of its operations. Analytical testing by a third party makes sure that product specifications are met independently. Manufacturers you can trust will gladly give you Certificates of Analysis from approved labs that show HPLC purity testing, heavy metal screening, microbial contamination analysis, and residue solvent testing. To check for stability across production runs, ask for more than one batch report instead of a single sample. Changes in test results could mean that the process controls aren't working right or that the quality of the raw materials isn't reliable. Setting clear acceptance criteria before starting a relationship with a supplier stops disagreements and lets you make objective quality assessments. Regulatory compliance documentation varies by target market but usually includes stability study data, manufacturing process descriptions, and safety assessment dossiers. Suppliers that do business in the U.S. should show that they know about FDA rules, such as the standards for dietary ingredient notification. To follow the rules for the European market, you need to know about the EFSA standards and, if necessary, the Novel Food regulations. Being clear about a product's regulatory state protects buyers from compliance risks and speeds up the registration process.
Conducting Effective Supplier Audits
Facility inspections, whether done virtually or in person, show operational skills that can't be fully captured by paper records. During audit trips, pay attention to how things are handled, how equipment is maintained, and how employees are trained. Well-run facilities have well-organised processes, good environmental controls, and thorough systems for keeping records. Some red flags are not enough space between production areas, bad housekeeping, or not wanting to answer scientific questions about how things are made. When evaluating a facility, the quality control laboratory should be given extra attention. HPLC devices for testing purity, moisture analysers for checking hydration, and spectroscopy tools for confirming identity should all be kept on hand for in-house analytical testing. The use of calibration records and method validation paperwork shows that analytical methods are scientifically sound. Suppliers who don't have the right testing tools may depend too much on outside labs, which could delay the release of batches and make quality control less effective. Traceability in the supply chain goes beyond the immediate manufacturer and includes sources of raw materials. For enzyme-based production methods to work, they need certain precursor substances and enzyme preparations, and each has its own quality requirements. Knowing the whole supply chain helps you figure out how vulnerable you are to problems and make sure that promises about sustainability are true. Transparent suppliers are happy to talk about how they get their raw materials and keep up with buyer qualification programs for important inputs.
Optimising Logistics and Inventory Management
When hygroscopic materials are shipped internationally, they need special packing and environmental controls. Aluminium foil laminate bags that are flushed with nitrogen keep wetness out during transport. Secondary packing in fibre drums adds mechanical protection while keeping unit sizes small enough to handle in the warehouse. In some markets or climates, temperature-controlled shipping may be necessary. This adds to the cost but makes sure the product arrives in good shape. Storage conditions have a big effect on stability and shelf life. Specifications usually say to store them below 25°C in places with low humidity and out of direct sunlight. Because of these needs, warehouse rooms need to be climate-controlled, especially in humid areas. Following the first-in, first-out rule for inventory movement keeps things from being stored for too long, which could affect their effectiveness. Setting maximum inventory age limits that are in line with stability data protects the quality of the product throughout the entire distribution chain. Minimum order numbers can be hard for new brands or product line extensions that don't know how much demand there will be. During the initial talks, negotiating flexible buying terms sets realistic goals and finds suppliers who are willing to work with growth-stage partners. Some makers offer inventory consignment plans or staged deliveries that lower the amount of money that needs to be paid up front while still making sure that there is a steady supply. When these creative business structures are put into action with clear decisions about quality standards and stable prices, they are good for everyone.
Building Trust and Making Informed Decisions as a B2B Buyer
Long-term procurement relationships depend on trust. Pure NMN Powder is built through consistent performance and transparent communication. The complexity of bioactive ingredient markets demands sophisticated evaluation frameworks that look beyond simple price comparisons.
Verifying Product Authenticity and Supplier Transparency
Analytical testing by unbiased third-party labs is an objective way to check what a provider says. Sending random samples to approved testing facilities gets rid of any possible bias and makes sure that the product meets the standards. When you compare results from different batches, you can see patterns of consistency that show that the factory controls are working well. There should be a thorough study into any discrepancies between a supplier's certificates and the results of independent tests. Unreliable partners may be ruled out. Suppliers who are honest about the manufacturing process are different from traders who are only looking to make money. Companies that invest in their own enzymatic synthesis technologies are usually more open to talking about production methods than traders who sell common materials. Visits to the sites that show advanced fermentation facilities and quality control labs show technical know-how and vertical integration. If a supplier doesn't want to give you access to their facilities or specific process descriptions, they may not have direct control over the manufacturing process. Intellectual property portfolios are an indirect way to show that you are technically sophisticated and committed to innovation. There are probably active research and development projects going on if you see patents for synthesis methods, purification techniques, or ways to make things more stable. Publications in peer-reviewed journals show that you are knowledgeable in your field and involved with the larger research group. In crowded markets, these factors help separate the real innovators from the suppliers of basic goods.
Key Performance Indicators for Supplier Partnerships
The dependability of deliveries has a direct effect on planning output and managing inventory. Track the percentage of on-time deliveries, the consistency of wait times, and how quickly you respond to urgent requests. Superior sellers keep extra stock on hand in case of sudden increases in demand and let customers know ahead of time about any delays that might happen. Setting up service level agreements with clear metrics lets you objectively evaluate performance and encourages ongoing improvement. Technical support goes beyond just processing orders; it also includes formulation advice, regulatory consultation, and help with fixing problems. During the development stages of a product, suppliers with specific technical service teams are very helpful. More value is added than just the physical ingredient itself when you can access stability data, formulation case studies, and market information. The extra cost of these support services is usually worth it because they cut down on development times and increase the chances of success. For strategic ingredients that are hard to get elsewhere, the financial health of the supplier organisations should be taken into account. Checking credit reports, financial statements, and the image of an industry can help you figure out the risks to business continuity. When suppliers are having money problems, they might cut corners on quality to save money or just quit the market without warning, which would mess up established supply lines. Diversifying your business across a number of qualified suppliers protects you in case one of them fails, while keeping prices and service under pressure from other suppliers.
support goes beyond just processing orders; it also includes formulation advice, regulatory consultation, and help with fixing problems. During the development stages of a product, suppliers with specific technical service teams are very helpful. More value is added than just the physical ingredient itself when you can access stability data, formulation case studies, and market information. The extra cost of these support services is usually worth it because they cut down on development times and increase the chances of success. For strategic ingredients that are hard to get elsewhere, the financial health of the supplier organisations should be taken into account. Checking credit reports, financial statements, and the image of an industry can help you figure out the risks to business continuity. When suppliers are having money problems, they might cut corners on quality to save money or just quit the market without warning, which would mess up established supply lines. Diversifying your business across a number of qualified suppliers protects you in case one of them fails, while keeping prices and service under pressure from other suppliers.
Conclusion
Nicotinamide mononucleotide has established itself as a cornerstone ingredient in the longevity supplement category, backed by compelling biochemical rationale and growing clinical evidence. Procurement success requires systematic evaluation of supplier capabilities, rigorous quality verification, and strategic relationship building with manufacturing partners. The technical complexity of this bioactive ingredient demands sophisticated buyers who understand the interplay between purity specifications, production methods, stability considerations, and regulatory compliance requirements. Organisations investing in comprehensive supplier qualification processes position themselves for sustainable competitive advantage through superior product quality and reliable supply continuity.
FAQ
1. What distinguishes Pure NMN Powder from standard nicotinamide products?
The fundamental difference lies in molecular structure and metabolic pathway utilisation. Standard nicotinamide enters NAD+ synthesis through rate-limited enzymatic steps involving NAMPT, creating metabolic bottlenecks. Nicotinamide mononucleotide bypasses these constraints by entering the salvage pathway downstream, enabling more efficient NAD+ restoration. Purity specifications also differ dramatically, with pharmaceutical-grade material achieving assay values exceeding 99% compared to lower-grade alternatives containing significant impurities.
2. How should manufacturers assess safety considerations for high-dose formulations?
Published clinical trials provide the evidence base for dosage safety assessments. Studies exploring intake levels up to 1000mg daily over periods extending to 12 weeks have documented favourable safety profiles with minimal adverse events. Manufacturers should review the complete clinical literature, considering subject demographics and monitoring parameters when establishing dosage recommendations. Consulting with regulatory affairs specialists helps navigate jurisdiction-specific requirements around dosage limits and safety substantiation.
3. What methods reliably authenticate supplier quality claims?
Independent third-party testing through accredited analytical laboratories provides the most reliable verification approach. HPLC analysis confirms purity specifications and detects adulterants or substitutions. Nuclear magnetic resonance spectroscopy offers additional confirmation of molecular identity and stereochemistry. Comparing test results across multiple suppliers and batches reveals quality consistency patterns. Facility audits supplement analytical testing by verifying manufacturing capabilities and quality systems implementation.
Partner with Angelbio for Your Pure NMN Powder Supply Needs
Angelbio brings together 18 years of independent research capability with advanced manufacturing infrastructure to serve as your trusted Pure NMN Powder supplier. Our joint investment partnership with Angel Holding Group and the Institute of Life and Health Research of Xi'an Jiaotong University delivers pharmaceutical-grade ingredients meeting the most stringent international quality standards. We maintain comprehensive testing protocols, GMP-certified production facilities, and global logistics networks designed specifically for nutraceutical procurement professionals.
Whether you're formulating next-generation longevity supplements, developing functional food applications, or expanding existing product lines, our technical team provides Pure NMN Powder customised support throughout your development process. We understand the unique challenges facing R&D directors and procurement managers navigating complex quality requirements and regulatory landscapes. Contact our team at angel@angelbiology.com to discuss your specific requirements, request analytical specifications, or arrange sample evaluation. Discover why leading brands choose Angelbio as their preferred Pure NMN Powder manufacturer for consistent quality, technical expertise, and dependable supply chain performance.
References
1. Yoshino, J., Mills, K. F., Yoon, M. J., & Imai, S. (2011). Nicotinamide mononucleotide, a key NAD+ intermediate, treats the pathophysiology of diet- and age-induced diabetes in mice. Cell Metabolism, 14(4), 528-536.
2. Mills, K. F., Yoshida, S., Stein, L. R., Grozio, A., Kubota, S., Sasaki, Y., & Imai, S. (2016). Long-term administration of nicotinamide mononucleotide mitigates age-associated physiological decline in mice. Cell Metabolism, 24(6), 795-806.
3. Poddar, S. K., Sifat, A. E., Haque, S., Nahid, N. A., Chowdhury, S., & Mehedi, I. (2019). Nicotinamide mononucleotide: exploration of diverse therapeutic applications of a potential molecule. Biomolecules, 9(1), 34.
4. Shade, C. (2020). The science behind NMN-A stable, reliable NAD+ activator and anti-aging molecule. Integrative Medicine: A Clinician's Journal, 19(1), 12-14.
5. Hong, W., Mo, F., Zhang, Z., Huang, M., & Wei, X. (2020). Nicotinamide mononucleotide: A promising molecule for therapy of diverse diseases by targeting NAD+ metabolism. Frontiers in Cell and Developmental Biology, 8, 246.
6. Igarashi, M., Nakagawa-Nagahama, Y., Miura, M., Kashiwabara, K., Yaku, K., Sawada, M., & Nakagawa, T. (2022). Chronic nicotinamide mononucleotide supplementation elevates blood nicotinamide adenine dinucleotide levels and alters muscle function in healthy older men. npj Aging, 8(1), 5.