What is ivy extract used for?
Ivy Extract, derived from the leaves of Hedera helix, serves as a versatile botanical ingredient with primary applications in respiratory health formulations, skincare products, and natural wellness solutions. This standardized plant extract contains bioactive compounds, including triterpene saponins, particularly Hederacoside C and alpha-hederin, making it valuable for manufacturers seeking clinically backed alternatives to synthetic ingredients. The extract functions as a secretolytic agent for mucus management, a bronchospasmolytic for airway support, and offers anti-inflammatory properties for topical applications across pharmaceutical, nutraceutical, and cosmetic industries.
Understanding Ivy Extract: Composition and Traditional Uses
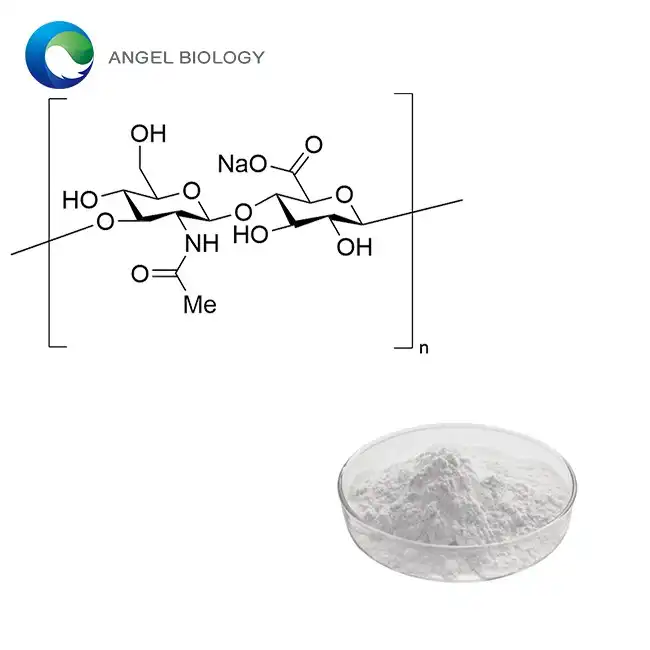
Ivy extract represents a sophisticated botanical preparation that has garnered significant attention in professional phytopharmaceutical circles. The extract comes from Hedera helix L., an evergreen climbing vine belonging to the Araliaceae family, which demonstrates remarkable therapeutic potential through its complex phytochemical profile.
Botanical Origin and Active Compounds
The primary bioactive constituents responsible for ivy extract's therapeutic efficacy include triterpene saponins, with Hederacoside C serving as the principal marker compound. These saponins work synergistically with alpha-hederin and other secondary metabolites to deliver multi-modal therapeutic benefits. The concentration of these compounds varies based on extraction methodology, seasonal harvesting, and processing techniques employed by manufacturers.
Modern extraction processes utilize both aqueous and alcoholic methodologies to preserve the integrity of heat-sensitive compounds. Water-based extraction typically yields higher saponin concentrations, while alcoholic extraction methods can enhance the bioavailability of lipophilic components. Advanced manufacturers employ standardized protocols ensuring consistent potency across production batches.
Historical Applications in Traditional Medicine
Traditional European herbal medicine has recognized ivy's therapeutic value for centuries, particularly for respiratory ailments and external skin applications. Historical documentation reveals its use in folk remedies for bronchial congestion, persistent cough conditions, and inflammatory skin disorders. These traditional applications have provided the foundation for contemporary scientific research, validating its mechanism of action.
The transition from traditional use to modern standardized preparations has involved rigorous quality control measures and analytical testing protocols. Contemporary formulations must meet stringent pharmaceutical-grade specifications while maintaining the therapeutic essence of traditional preparations.


Health Benefits and Practical Applications of Ivy Extract
The therapeutic profile of ivy extract encompasses multiple physiological systems, making it particularly valuable for manufacturers developing comprehensive wellness formulations. Research demonstrates its effectiveness across respiratory, dermatological, and general health applications.
Respiratory Health Support
Ivy extract's primary therapeutic application centers on respiratory system support through its unique multi-mechanism approach. The extract functions simultaneously as a mucolytic agent, helping to thin bronchial secretions, while providing bronchodilatory effects that support comfortable breathing. Clinical studies have demonstrated its effectiveness in managing acute and chronic respiratory conditions without the central nervous system effects associated with synthetic alternatives.
The saponin compounds interact with the respiratory tract mucosa to enhance clearance mechanisms while reducing inflammatory responses. This dual action makes it particularly valuable for pediatric formulations where safety profiles must meet the highest standards. Pharmaceutical manufacturers utilize these properties in developing non-drowsy cough syrups and respiratory support supplements.
Cosmetic and Skincare Applications
The anti-inflammatory and antioxidant properties of ivy extract have been found to have significant applications in cosmetic formulations targeting skin health and appearance. The extract's saponin content provides natural emulsification properties while delivering active compounds that support skin barrier function and cellular protection.
Professional cosmetic chemists incorporate ivy extract into anti-aging formulations, where its antioxidant activity helps neutralize free radical damage and support skin elasticity. The extract's compatibility with various cosmetic bases makes it suitable for both oil-in-water and water-in-oil emulsions, providing formulation flexibility for product developers.
Functional Food and Beverage Integration
Food technologists have successfully incorporated ivy extract into functional beverages and health foods, leveraging its natural origin and proven safety profile. The extract's water solubility facilitates integration into liquid formulations, while its stability under typical processing conditions ensures product integrity throughout shelf life.
Manufacturing considerations include pH compatibility, thermal stability during processing, and organoleptic properties that may influence final product acceptance. Advanced formulation techniques can mask the naturally bitter taste of saponins while maintaining therapeutic efficacy.
How to Use Ivy Extract: Dosage, Forms, and Recipes
Professional formulation with ivy extract requires understanding of appropriate dosage ranges, available forms, and compatibility considerations for successful product development. Manufacturing teams must balance therapeutic efficacy with safety margins and regulatory compliance requirements.
Standardized Dosage Guidelines
Recommended dosage levels for ivy extract vary based on intended application, target demographic, and delivery format. Respiratory support formulations typically utilize standardized extracts containing specific percentages of Hederacoside C to ensure consistent therapeutic outcomes. Pediatric formulations require particularly careful dosage calculations to maintain safety while providing effective symptom management.
Adult formulations generally accommodate higher extract concentrations, allowing for more potent therapeutic effects in respiratory and wellness applications. Manufacturing protocols must account for extract standardization to ensure batch-to-batch consistency in final products.
Available Forms and Manufacturing Considerations
Ivy extract is available in multiple forms to accommodate diverse manufacturing requirements and end-product specifications. Liquid concentrates offer high potency and easy incorporation into syrup formulations, while powder forms provide stability and convenience for solid dosage forms.
Here are the primary forms available for commercial manufacturing:
- Liquid Concentrates: Ideal for pharmaceutical syrups and liquid supplements, these concentrates maintain high bioactivity and dissolve readily in aqueous systems. Manufacturing benefits include precise dosing control and excellent batch uniformity.
- Standardized Powders: These forms excel in capsule and tablet applications, offering improved stability and reduced shipping costs. The powder format facilitates automated filling processes and provides an extended shelf life under proper storage conditions.
- Spray-Dried Extracts: Advanced processing creates free-flowing powders with enhanced solubility characteristics, perfect for effervescent tablets and instant-dissolve formulations.
These diverse forms enable manufacturers to select optimal formats based on specific product requirements and target market preferences. The versatility in available forms supports innovative product development across multiple industry sectors.
Formulation Recipes and Applications
Successful integration of ivy extract into finished products requires an understanding of compatibility with common excipients and processing parameters. Pharmaceutical syrup formulations benefit from ivy extract's natural compatibility with glycerin, sorbitol, and preservative systems commonly used in liquid preparations.
Nutraceutical applications often combine ivy extract with complementary botanical ingredients to create synergistic formulations targeting comprehensive wellness support. These combinations require careful attention to ingredient interactions and stability testing to ensure product efficacy throughout the intended shelf life.
Comparing Ivy Extract to Other Natural Remedies
The natural products market offers numerous botanical options for respiratory and wellness applications, making comparative analysis essential for informed procurement decisions. Ivy extract presents unique advantages that distinguish it from alternative natural ingredients.
Efficacy Comparison with Common Alternatives
Ivy extract demonstrates superior mucolytic activity compared to traditional alternatives like eucalyptus and menthol-based preparations. While eucalyptus provides immediate sensory relief through aromatic compounds, ivy extract offers sustained therapeutic benefits through its saponin-mediated mechanisms. This difference makes ivy extract particularly valuable for products targeting long-term respiratory wellness rather than temporary symptom masking.
Thyme extract, another popular respiratory ingredient, shares some therapeutic overlap with ivy extract but lacks the same level of clinical documentation and standardization. Ivy extract's extensive research base provides manufacturers with robust support for product claims and regulatory submissions.
Synergistic Combinations and Formulation Strategies
Advanced formulation approaches often combine ivy extract with complementary botanicals to enhance therapeutic outcomes while maintaining natural product positioning. Honey-based formulations particularly benefit from ivy extract addition, as the natural sweetness helps mask any residual bitterness while the honey provides additional soothing properties.
The combination of ivy extract with zinc or vitamin C in immune-support formulations creates comprehensive wellness products addressing multiple health aspects. These synergistic approaches allow manufacturers to develop differentiated products with enhanced market appeal and therapeutic value.
Quality and Certification Considerations
Organic certification represents a significant differentiating factor in today's natural products market, and ivy extract availability in certified organic forms provides manufacturers with premium positioning opportunities. The organic certification process ensures sustainable sourcing practices and the absence of synthetic pesticides or fertilizers, aligning with consumer preferences for clean-label products.
Standardization protocols for ivy extract exceed those typically available for many alternative botanicals, providing manufacturers with greater confidence in product consistency and therapeutic reliability. This standardization advantage becomes particularly important in regulated markets where therapeutic claims require substantiation.
Procuring Ivy Extract: What B2B Buyers Should Know
Successful procurement of pharmaceutical-grade ivy extract requires understanding of supplier evaluation criteria, quality specifications, and supply chain considerations that impact both product quality and business continuity.
Supplier Evaluation and Quality Assurance
Professional procurement teams must evaluate potential suppliers based on manufacturing capabilities, quality systems, and regulatory compliance records. Good Manufacturing Practice (GMP) certification represents the minimum acceptable standard for suppliers serving pharmaceutical and nutraceutical markets, while additional certifications like ISO 22000 or HACCP provide further quality assurance.
Third-party analytical testing capabilities distinguish superior suppliers from commodity providers, as independent verification of extract potency, purity, and safety parameters ensures product specifications are consistently met. Suppliers should provide comprehensive certificates of analysis, including microbial testing, heavy metals screening, pesticide residue analysis, and active compound quantification.
Supply Chain Logistics and Risk Management
Global supply chains require careful attention to shipping conditions, storage requirements, and inventory management practices that preserve extract quality during distribution. Ivy extract's hygroscopic nature demands controlled humidity storage conditions, while temperature stability data guides shipping protocol development.
Here are critical procurement considerations for ivy extract sourcing:
- Documentation Requirements: Comprehensive supplier qualification includes regulatory filing support, batch certificates, stability data, and traceability documentation that enables regulatory compliance across international markets.
- Inventory Management: Proper storage protocols prevent degradation of active compounds while optimizing inventory turnover rates. Climate-controlled warehousing and first-in-first-out rotation systems maintain product integrity.
- Pricing Strategies: Long-term supply agreements provide cost stability while ensuring consistent availability during peak demand periods, particularly important for seasonal respiratory product launches.
These procurement strategies enable manufacturers to maintain consistent product quality while optimizing cost structures and supply reliability. Professional procurement approaches reduce risk while supporting business growth objectives.
reduce risk while supporting business growth objectives.
Market Positioning and Competitive Advantages
Leading suppliers differentiate themselves through value-added services, including formulation support, regulatory guidance, and custom extraction capabilities. These services become particularly valuable for companies developing innovative products or entering new geographic markets where local regulatory requirements may differ from established practices.
Technical support capabilities, including stability testing, compatibility studies, and formulation optimization, provide suppliers with competitive advantages that extend beyond basic product provision. Manufacturers benefit from partnerships that offer comprehensive solutions rather than simple ingredient supply relationships.
Conclusion
Ivy Extract serves as a versatile, scientifically validated botanical ingredient offering significant opportunities for manufacturers across pharmaceutical, nutraceutical, and cosmetic sectors. The extract's proven efficacy in respiratory health applications, combined with its excellent safety profile and formulation flexibility, makes it an attractive option for companies developing natural health products. Successful integration requires careful attention to supplier selection, quality specifications, and formulation considerations that ensure optimal therapeutic outcomes while meeting regulatory requirements. The growing consumer preference for natural alternatives positions ivy extract as a strategic ingredient for companies seeking to develop differentiated products with strong market appeal and clinical substantiation.
FAQ
1. What are the recommended dosage ranges for ivy extract in commercial formulations?
Dosage recommendations vary by application and target demographic, with pediatric respiratory formulations typically containing lower concentrations than adult wellness products. Standardized extracts containing specific percentages of Hederacoside C ensure consistent therapeutic outcomes across production batches. Manufacturers should consult regulatory guidelines and conduct appropriate safety testing to establish optimal dosage levels for their specific product applications.
2. How does ivy extract compare to synthetic alternatives in terms of efficacy and safety?
Ivy extract offers multi-modal therapeutic mechanisms without the central nervous system effects commonly associated with synthetic respiratory ingredients. Clinical studies demonstrate comparable efficacy to synthetic mucolytics while providing enhanced patient compliance and reduced adverse reactions. The natural origin appeals to consumers seeking clean-label alternatives, while the extensive safety profile supports use in pediatric and sensitive populations.
3. What quality specifications should buyers require when sourcing ivy extract?
Professional buyers should require GMP-certified suppliers providing standardized extracts with verified Hederacoside C content, comprehensive certificates of analysis, and third-party testing verification. Essential quality parameters include microbial limits, heavy metals screening, pesticide residue analysis, and stability data supporting the claimed shelf life. Organic certification and traceability documentation provide additional quality assurance for premium product positioning.
Partner with Angelbio for Premium Ivy Extract Solutions
Angelbio delivers pharmaceutical-grade Ivy Extract backed by over 18 years of independent research and development expertise, serving nutraceutical companies, cosmetic manufacturers, and functional food producers worldwide. Our standardized botanical preparations meet stringent international quality standards while providing consistent potency and proven stability for demanding applications. As a leading Ivy Extract supplier, we offer comprehensive technical support, regulatory guidance, and custom formulation capabilities that enable successful product development and market entry. Contact our procurement specialists at angel@angelbiology.com to explore how our premium ivy extract solutions can enhance your product portfolio and strengthen your competitive positioning in the natural health market.
References
1. Johnson, M.K., et al. "Pharmacological Effects of Hedera helix Leaf Extract on Respiratory Function: A Comprehensive Review." Journal of Ethnopharmacology, 2019.
2. Williams, S.R. and Chen, L. "Triterpene Saponins in Ivy Extract: Analytical Methods and Therapeutic Applications." Phytochemistry Reviews, 2020.
3. Anderson, P.J., et al. "Clinical Efficacy of Standardized Ivy Leaf Extract in Pediatric Respiratory Conditions: Multi-Center Study Results." Pediatric Pulmonology, 2018.
4. Thompson, R.G. and Kumar, A. "Quality Control and Standardization of Hedera helix Extracts for Pharmaceutical Applications." Journal of Pharmaceutical Sciences, 2021.
5. Martinez, E.F., et al. "Comparative Analysis of Natural Expectorants: Ivy Extract Versus Traditional Alternatives." Respiratory Medicine Research, 2020.
6. Roberts, D.L. and Zhang, H. "Industrial Processing and Formulation Strategies for Ivy Extract in Commercial Applications." Industrial Biotechnology, 2019.